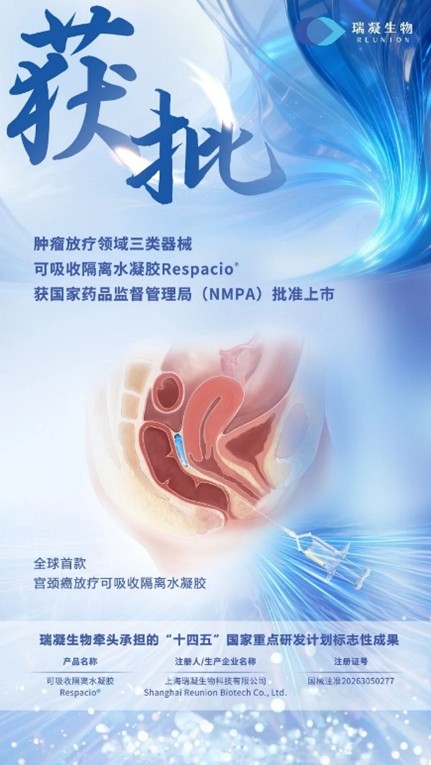
Recently, Respacio®, an absorbable isolation hydrogel for tumor radiotherapy independently developed by Shanghai Reunion Biotechnology Co., Ltd. (hereinafter referred to as "Reunion"), has been approved for market launch by the Chinese National Medical Products Administration (NMPA). Respacio® is the landmark achievement of the key special project "Medical Equipment and Biological Medical Materials" under the National Key R&D Program of the 14th Five-Year Plan, led by Reunion. It is also the world's first approved isolation hydrogel product specifically designed for cervical cancer radiotherapy, which will facilitate a leapfrog advancement in the field of domestic radiotherapy. Tigermed-Jyton provided full-process clinical services for the successful market launch of this product.
(Image source: Reunion WeChat official account)
First-in-class Isolation Gel, Injecting Technological Warmth Into Precision Radiotherapy
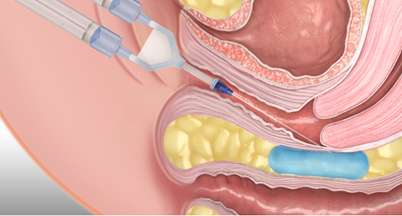
In China, cervical cancer has a high incidence rate, and radiotherapy plays a crucial role in its treatment. Although the 5-year survival rate after radiotherapy can reach 70%, improved survival has not been fully translated into better quality of life, and complications such as radiation-induced rectal injury remain clinical challenges. Reunion, supported by a founding team originating from top biomaterials laboratories in China and the United States, has successfully developed Respacio® absorbable separation hydrogel based on an in-house developed solution-stable polyethylene glycol (PEG) hydrogel platform. This product is precisely implanted between the tumor and the rectum via minimally invasive injection. By leveraging optimized polymer molecular configurations and innovative cross-linking technology, it forms a separation layer with excellent conformability, stability, and multi-modal visibility, significantly reducing radiation dose to the rectum and adjacent healthy tissues, thereby effectively mitigating radiation-induced rectal injury. The emergence of Respacio® provides a novel organ protection strategy for patients undergoing repeated radiotherapy, offering the potential not only to reduce treatment-related side effects and markedly improve quality of life, but also to shorten radiotherapy duration and optimize treatment pathways, driving the evolution of conventional radiotherapy toward greater precision and patient-centric care.
(Image source: Reunion official website)
Prospective Clinical Strategy,Paving the Way for Innovative Device Translation
As the world's first hydrogel designed for rectal spacer in cervical cancer radiotherapy, the clinical trial and registration pathway for Respacio® had no precedents. Facing a complete absence of referenceable cases and literature at the initial stage of protocol design, the Tigermed-Jyton Oncology Project Team leveraged its profound expertise in the field of radiotherapy and worked closely with the sponsor to innovatively develop a clinical trial protocol that met both scientific and regulatory requirements. In terms of registration strategy, the team proactively planned for an extended follow-up period to collect more comprehensive long-term safety data, laying a solid foundation for successful product approval. To ensure study quality, the project introduced an expert panel to professionally review and strictly oversee patients' radiotherapy protocols, ensuring standardized procedures and rigorous, reliable data. From the ground-up protocol design to forward-looking strategic planning and professional study management, Tigermed-Jyton delivered end-to-end clinical services that successfully addressed the challenges of translating innovative medical devices from "0 to 1".
The successful market launch of Respacio® marks not only a milestone for Reunion but also another benchmark case for Tigermed-Jyton in the field of innovative oncology medical devices. The Tigermed-Jyton oncology device team is a specialized team focused on the oncology field, with team members possessing extensive experience in oncology. They have accumulated solid expertise in product areas such as radiotherapy equipment, electric field therapy, and interventional therapy, covering multiple departments including neurosurgery, urology, gynecology, respiratory, thoracic surgery, gastroenterology, pancreatology, and interventional radiology. Tigermed-Jyton has always been committed to providing global medical device companies with comprehensive lifecycle services, including product remediation, strategic consulting, quality system guidance, clinical trials, registration submissions, and post-market support. Going forward, Tigermed-Jyton will continue to leverage its keen policy insights and professional technical team to collaborate with medical device companies, enabling more innovative medical device products to benefit patients worldwide.
About Reunion

Shanghai Reunion Biotechnology Co., Ltd. is a ‘specialized and sophisticated enterprises that produce new and unique products’, focusing on the research, development and commercialization of high-end biomedical materials. Founded by global education background PhDs, the company is a high-tech company dedicated to the commercialization of state-of-the-art biomaterials. The company has built the world‘s leading medical hydrogel technology platform to provide innovative medical device solutions for oncology, and established Reunion’s unique and world‘s first “ready-to-use” PEG hydrogel platform.
Reunion Biotech has a footage of more than 4000 m2, including R&D center, GMP facilities and physical, chemical and microbiological testing laboratories . The company is comprised of top talents in product development, quality control and corporate management. The company has received hundreds of millions RMB investment from Qiming Venture Capital and other famous domestic and foreign investment organizations. Meanwhile, the company has established in-depth cooperation with universities, research institutes and top tertiary hospitals to develop innovative products.